Learning to draw Lewis structures, especially for ions like CN-, can feel like a puzzle, yet it's a fundamental skill in chemistry that helps us see how atoms connect and share electrons. It's almost like figuring out the blueprint for a tiny molecular building. Understanding the cyanide ion's structure, a compound known for its potent nature, really shows us the core principles of chemical bonding in action. This guide will walk you through each step, making sure you grasp not just the "how" but also a bit of the "why" behind creating this important diagram.
Many students, you know, find themselves wondering about the best way to approach these structures. It's a common query, actually, whether you're just starting out in middle school chemistry or tackling more advanced topics in college. Our goal here is to give you a clear, easy-to-follow path, drawing on established methods and examples, so you can confidently sketch out the CN- Lewis structure and, in fact, many other similar chemical diagrams.
So, we'll cover everything from counting those all-important valence electrons to figuring out the right arrangement of atoms and electrons. We will, of course, make sure to talk about formal charges, which are pretty important for checking if your structure is the most favorable one. Let's get started on figuring out the CN- Lewis structure, a rather key part of understanding covalent compounds.
- Melissa Peterman Height
- Jude Bellingham Religion
- Did Celine Dion Son Get Married
- Who Is The Mother Of Yannis Daughter
- Remoteiot Vpc Ssh Raspberry Pi Aws Example
Table of Contents
- What Exactly is a Lewis Structure?
- The Cyanide Ion (CN-): A Closer Look
- Step-by-Step Guide to Drawing the CN- Lewis Structure
- Molecular Geometry and Stability of CN-
- Common Challenges and Tips
- Frequently Asked Questions About CN- Lewis Structure
What Exactly is a Lewis Structure?
A Lewis structure, sometimes called a Lewis dot formula, is a really handy diagram that shows us how atoms connect and share electrons when they form a molecule or an ion. It's a visual way to represent the valence electrons, which are the electrons in the outermost shell of an atom, and these are the ones that actually participate in chemical bonding. Basically, you know, it simplifies the whole quantum mechanical picture of atoms into something we can easily draw and understand.
These structures are pretty important because they help us predict a molecule's shape, its reactivity, and other chemical properties. They show us all of the valence electrons, both those involved in bonds and those that are just sitting there as "lone pairs." Bonds are usually shown as lines between atoms, and lone pairs are shown as dots. It's a simple, yet powerful, tool for chemists, actually.
The main idea behind Lewis structures is often the octet rule, which says that most main group elements want to have eight valence electrons around them to achieve a stable electron configuration, much like the noble gases. Hydrogen, of course, is an exception, only needing two electrons. So, in some respects, Lewis structures are about helping atoms achieve this stable state by sharing electrons.
- Joe Wen Net Worth
- Daz Dillinger Net Worth 2024
- Raspberry Pi Remote Iot Download Free
- Ways To Say Many Level 71
- Sabrina Jackson
The Cyanide Ion (CN-): A Closer Look
The cyanide ion, written as CN-, is a rather interesting and, in fact, quite important chemical species. It consists of one carbon atom and one nitrogen atom connected together, carrying an overall negative charge. This negative charge, you see, is pretty crucial when we're counting valence electrons for our Lewis structure.
Cyanides are chemical compounds that, as a matter of fact, are fast-acting poisons. You might have heard of hydrogen cyanide (HCN) or salts like potassium cyanide (KCN) and sodium cyanide (NaCN), which are among the most dangerous forms. However, our focus here isn't on its toxicity, but purely on its structural representation in chemistry. It's just a little bit of background, you know.
Understanding the Lewis structure of CN- is, therefore, a key step in learning about covalent bonding and how different atoms come together to form stable ions. The linear arrangement of its atoms, as we will see, also tells us something about its stability and behavior in chemical reactions. It's a pretty fundamental example, actually, for many chemistry lessons.
Step-by-Step Guide to Drawing the CN- Lewis Structure
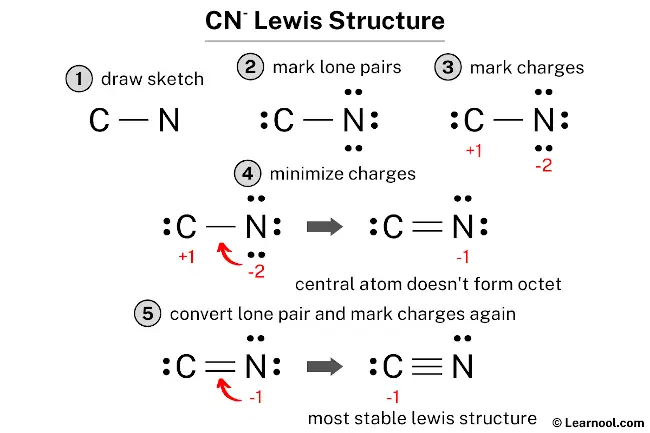
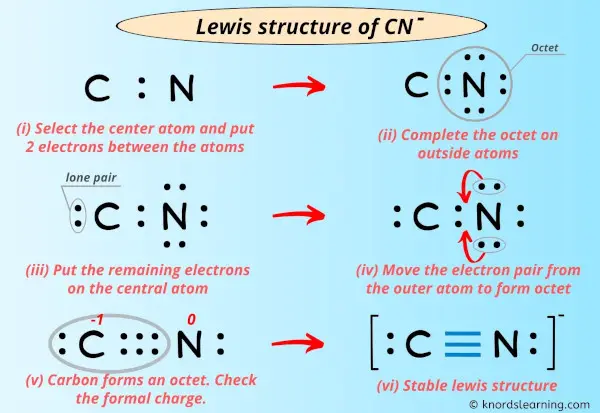
Drawing the Lewis structure for the cyanide ion (CN-) is, in a way, a straightforward process if you follow a set of clear steps. My text, for instance, mentions following a few steps with examples. Let's break it down, making sure we cover all the details to help you create an accurate diagram. This is, you know, the practical part.
Step 1: Count the Total Valence Electrons
The very first thing we need to do is figure out the total number of valence electrons available for bonding in the CN- ion. Carbon (C) and nitrogen (N) are both main group elements, so their group number tells us their valence electrons. Carbon, for example, is in Group 14, so it has 4 valence electrons. Nitrogen, on the other hand, is in Group 15, so it has 5 valence electrons. It's pretty simple to find these, really.
Since we are dealing with an ion that has a negative charge (CN-), that means there's an extra electron involved. A single negative charge means we add one more electron to our total count. So, for CN-, the calculation goes like this:
- Carbon (C): 4 valence electrons
- Nitrogen (N): 5 valence electrons
- Negative charge (-1): +1 electron
- Total valence electrons = 4 + 5 + 1 = 10 electrons
This total of 10 electrons is, in fact, what we have to work with to form bonds and lone pairs in our Lewis structure. It's quite a critical number, you know, to get right from the start.
Step 2: Arrange the Atoms
For a diatomic ion like CN-, arranging the atoms is actually pretty simple. There are only two atoms, carbon and nitrogen, so they must be connected directly to each other. There isn't, you know, a central atom in the way you might think of for a molecule with three or more atoms. They just sit side-by-side.
So, we'll place the carbon atom next to the nitrogen atom. It's a linear arrangement by default for just two atoms, which, in a way, makes this step very easy. You can just write them down as C-N, for instance, as a starting point. This initial setup is, really, the foundation for the rest of your drawing.
Step 3: Draw Single Bonds
Next, we need to draw a single bond between the carbon and nitrogen atoms. A single bond represents two shared electrons. This is the initial connection that holds the atoms together. So, we'll draw a line between C and N. This line, you know, uses up two of our total valence electrons.
Let's keep track of our electrons:
- Total valence electrons: 10
- Electrons used for one single bond: 2
- Remaining electrons: 10 - 2 = 8 electrons
We still have 8 electrons left to distribute around the atoms to satisfy their octets. This is, you know, a pretty standard procedure in Lewis structures.
Step 4: Distribute Remaining Electrons as Lone Pairs
Now, we take those remaining 8 electrons and distribute them as lone pairs around the atoms, trying to satisfy the octet rule for each atom. Typically, you start by placing lone pairs on the more electronegative atom first, which in this case is nitrogen. However, with only two atoms, we just aim to give both atoms an octet.
Let's try to give each atom 8 electrons (including the shared ones from the single bond). Each atom already has 2 electrons from the single bond. So, each needs 6 more electrons to complete its octet.
- If we give 6 electrons (3 lone pairs) to nitrogen: N will have 2 (from bond) + 6 (lone pairs) = 8 electrons.
- This leaves 8 - 6 = 2 electrons for carbon.
- If we give 2 electrons (1 lone pair) to carbon: C will have 2 (from bond) + 2 (lone pair) = 4 electrons.
At this point, nitrogen has an octet, but carbon only has 4 electrons. This is, you know, a common situation where we need to adjust things. My text mentions that sometimes there aren't enough valence electrons for each atom to achieve an octet with just single bonds and lone pairs, and this is exactly that kind of scenario.
Step 5: Form Multiple Bonds if Needed
Since carbon doesn't have an octet (it only has 4 electrons), we need to move some lone pair electrons from nitrogen to form additional bonds between carbon and nitrogen. This is how atoms can achieve their octets when there aren't enough electrons for lone pairs alone. We'll take lone pairs from nitrogen and convert them into shared bonding pairs.
Nitrogen currently has 3 lone pairs (6 electrons). Let's move one lone pair from nitrogen to form a double bond between C and N. This means we'll now have a double bond, using 4 shared electrons, and nitrogen will have 2 lone pairs left (4 electrons). Carbon will now have 4 (from double bond) + 2 (its own lone pair) = 6 electrons. Still not an octet for carbon, you see.
So, we need to move another lone pair. Let's take another lone pair from nitrogen and form a triple bond between C and N. Now, the structure looks like this:
- C ≡ N
Let's check the electrons again:
- The triple bond uses 6 shared electrons.
- Nitrogen had 3 lone pairs. We moved two of them to form the triple bond. So, nitrogen now has 1 lone pair (2 electrons).
- Carbon still has its 1 lone pair (2 electrons).
Now, let's count the electrons around each atom:
- For Carbon (C): 6 (from triple bond) + 2 (from its lone pair) = 8 electrons (octet satisfied!).
- For Nitrogen (N): 6 (from triple bond) + 2 (from its lone pair) = 8 electrons (octet satisfied!).
Both atoms now have a complete octet, and we've used all 10 valence electrons (6 in the triple bond + 2 on C + 2 on N = 10). This structure, you know, is looking pretty good.
Step 6: Calculate Formal Charges
Calculating formal charges is a very important step to confirm that we have the most favorable Lewis structure. The closer the formal charges are to zero, the more likely we have the most stable structure, as my text suggests. The formula for formal charge is:
Formal Charge = (Valence Electrons) - (Non-bonding Electrons) - (1/2 Bonding Electrons)
Let's apply this to our CN- structure (C≡N, with one lone pair on C and one lone pair on N):
For Carbon (C):
- Valence electrons for C: 4
- Non-bonding electrons on C (lone pair): 2
- Bonding electrons for C (from triple bond): 6 (since it shares 1/2 of the 6 electrons)
- Formal Charge (C) = 4 - 2 - (1/2 * 6) = 4 - 2 - 3 = -1
For Nitrogen (N):
- Valence electrons for N: 5
- Non-bonding electrons on N (lone pair): 2
- Bonding electrons for N (from triple bond): 6 (since it shares 1/2 of the 6 electrons)
- Formal Charge (N) = 5 - 2 - (1/2 * 6) = 5 - 2 - 3 = 0
The sum of the formal charges is -1 + 0 = -1, which matches the overall charge of the cyanide ion. This is, you know, a really good sign that our structure is correct. The carbon atom carries the negative charge in this most stable Lewis structure, which is consistent with nitrogen being slightly more electronegative and "pulling" electron density, leaving carbon with a formal negative charge. This structure follows the octet rule for both atoms, which is pretty stable.
So, the final Lewis structure for CN- looks like this:
[ :C≡N: ]⁻
Where the brackets and the negative sign indicate that it's an ion with an overall negative charge. This is, you know, the way we usually show ions in Lewis structures.
Molecular Geometry and Stability of CN-
When we talk about the molecular geometry of CN-, it's actually pretty straightforward because there are only two atoms. Any two atoms connected form a linear shape. This linear arrangement, you see, is inherently stable for a diatomic molecule or ion. My text, for example, mentions seeing examples and diagrams of linear structures and how this arrangement suggests high stability.
The bond angle in a linear molecule is 180 degrees, though for just two atoms, it's more about the straight line between them. This simple, direct connection, you know, minimizes electron-electron repulsion, contributing to its overall stability. The triple bond between carbon and nitrogen is also very strong, which, in a way, adds to the ion's stability.
This linear geometry is a direct result of the Lewis structure we've drawn. The presence of the triple bond and the lone pairs on each atom dictates this shape. It's a pretty clear connection between the electron arrangement and the physical shape of the ion, actually.
Common Challenges and Tips
Drawing Lewis structures can, at times, present a few challenges, especially for ions or molecules where multiple bonds are needed. One common issue, you know, is not having enough valence electrons to satisfy octets with just single bonds, as we saw with CN-. This is a clear signal that you'll need to start forming double or triple bonds by moving lone pairs into shared bonding positions.
Another thing people sometimes struggle with is correctly calculating formal charges. It's really important to remember that formal charge helps you pick the best structure among several possibilities. The structure with formal charges closest to zero, and with negative formal charges on the more electronegative atoms, is usually the most favorable one. You can, for instance, double-check your math every time.
Here are some quick tips:
- Always recount: After distributing electrons, always count the total electrons used and compare it to your initial total valence electrons. They must match.
- Check octets: Make sure every atom (except hydrogen, which needs 2) has 8 electrons around it. If not, consider multiple bonds.
- Practice makes perfect: The more you draw Lewis structures for various molecules and ions, like other interesting chemical structures, the easier it becomes. You'll start to recognize patterns.
- Use a systematic approach: Follow the steps consistently every time. This helps avoid missing any crucial part of the process.
- Think about electronegativity: When assigning formal charges, remember that negative charges are more stable on more electronegative atoms.
These strategies, you know, can really help you get better at this skill. It's just a matter of applying them consistently.
Frequently Asked Questions About CN- Lewis Structure
People often have similar questions when they're trying to figure out the CN- Lewis structure. Here are a few common ones, actually, that might help clarify things even further.
What is the total number of valence electrons in CN-?
The total number of valence electrons in the cyanide ion (CN-) is 10. Carbon contributes 4, nitrogen contributes 5, and the negative charge adds an additional electron. So, you know, 4 + 5 + 1 equals 10 electrons in total.
Is CN- a linear molecule?
Yes, CN- is a linear ion. With only two atoms, carbon and nitrogen, they naturally arrange themselves in a straight line. This linear arrangement, you see, is a pretty characteristic feature of diatomic species.
Why does carbon have a negative formal charge in the CN- Lewis structure?
Carbon ends up with a negative formal charge (-1) in the most stable Lewis structure for CN- because of the way electrons are shared in the triple bond and the lone pair on carbon. Even though nitrogen is more electronegative, the electron distribution required to satisfy the octet rule for both atoms, particularly carbon, leads to carbon formally holding the extra electron. It's just a consequence of the electron sharing, really.
For more detailed insights into how these structures relate to overall molecular behavior, you might want to explore more advanced topics on molecular bonding. It's a pretty big subject, actually.
- Who Is Geoffrey Thorne Wife
- Piper Presley Leak Of
- Himesh Patel Partner
- Jesse Morales Age
- Jay Roach Net Worth
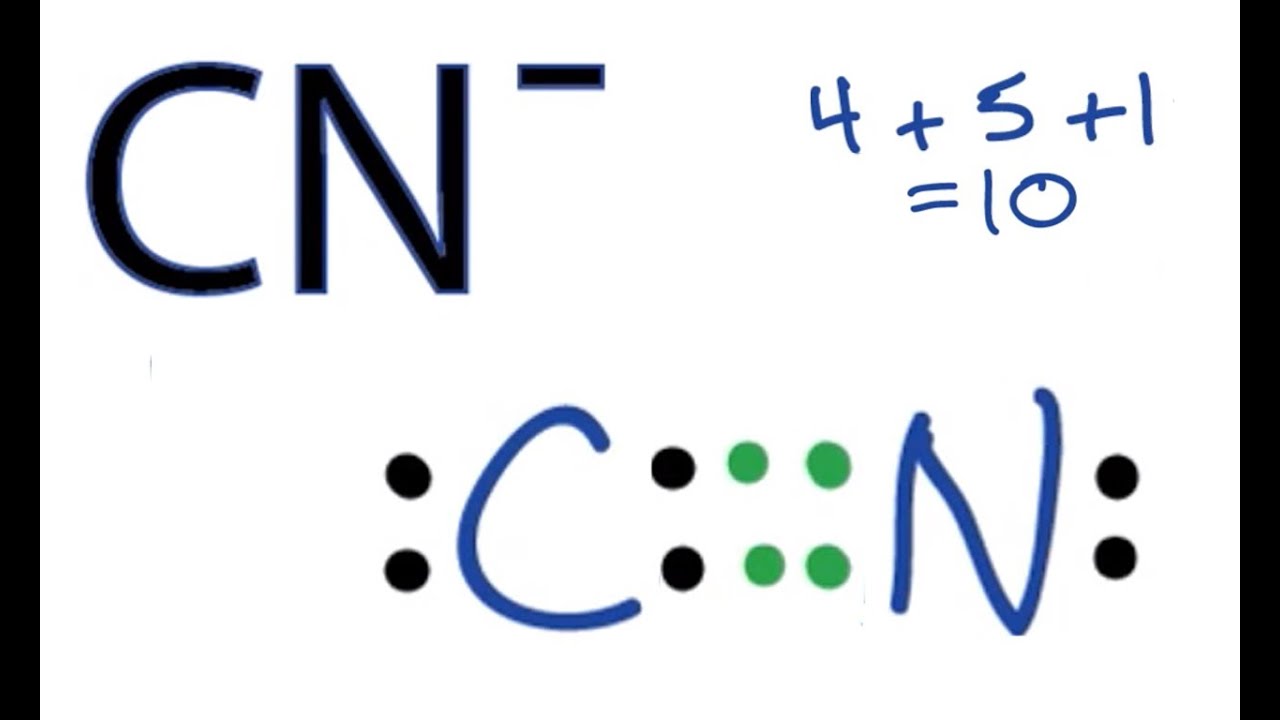
Detail Author:
- Name : Prof. Caitlyn Lindgren IV
- Username : dietrich.brown
- Email : rylan.runte@yahoo.com
- Birthdate : 1998-05-07
- Address : 8574 Ruthie Islands Noemyburgh, GA 31502
- Phone : 743-286-9233
- Company : Lehner, Little and Skiles
- Job : Tree Trimmer
- Bio : Qui aut blanditiis a qui unde consectetur excepturi. A tempora delectus eum qui. Cumque vitae in illum ex quisquam adipisci doloremque.
Socials
tiktok:
- url : https://tiktok.com/@mariane_hudson
- username : mariane_hudson
- bio : Dolor ut commodi minima. Aspernatur et vel laborum libero fugit.
- followers : 2455
- following : 87
facebook:
- url : https://facebook.com/hudsonm
- username : hudsonm
- bio : Inventore assumenda perferendis ab sit non est in.
- followers : 2120
- following : 2075
linkedin:
- url : https://linkedin.com/in/mhudson
- username : mhudson
- bio : Porro perferendis quis dicta minima et atque et.
- followers : 5639
- following : 2556
twitter:
- url : https://twitter.com/mariane_official
- username : mariane_official
- bio : Deserunt omnis consectetur veniam ab quos sint. Debitis repellat molestiae qui delectus qui temporibus totam. Et nulla nostrum quae recusandae assumenda qui.
- followers : 1347
- following : 829
instagram:
- url : https://instagram.com/mhudson
- username : mhudson
- bio : Quae aut in et explicabo quis. Sit iusto id magnam optio sequi quis.
- followers : 3692
- following : 2457