Ever wondered how atoms in a molecule arrange themselves, or why some ions behave the way they do? Well, getting a handle on the lewis structure of cn- is a pretty good place to start, actually. It's like peeking behind the curtain to see the electron dance that holds everything together, and it's a fundamental concept in chemistry, you know.
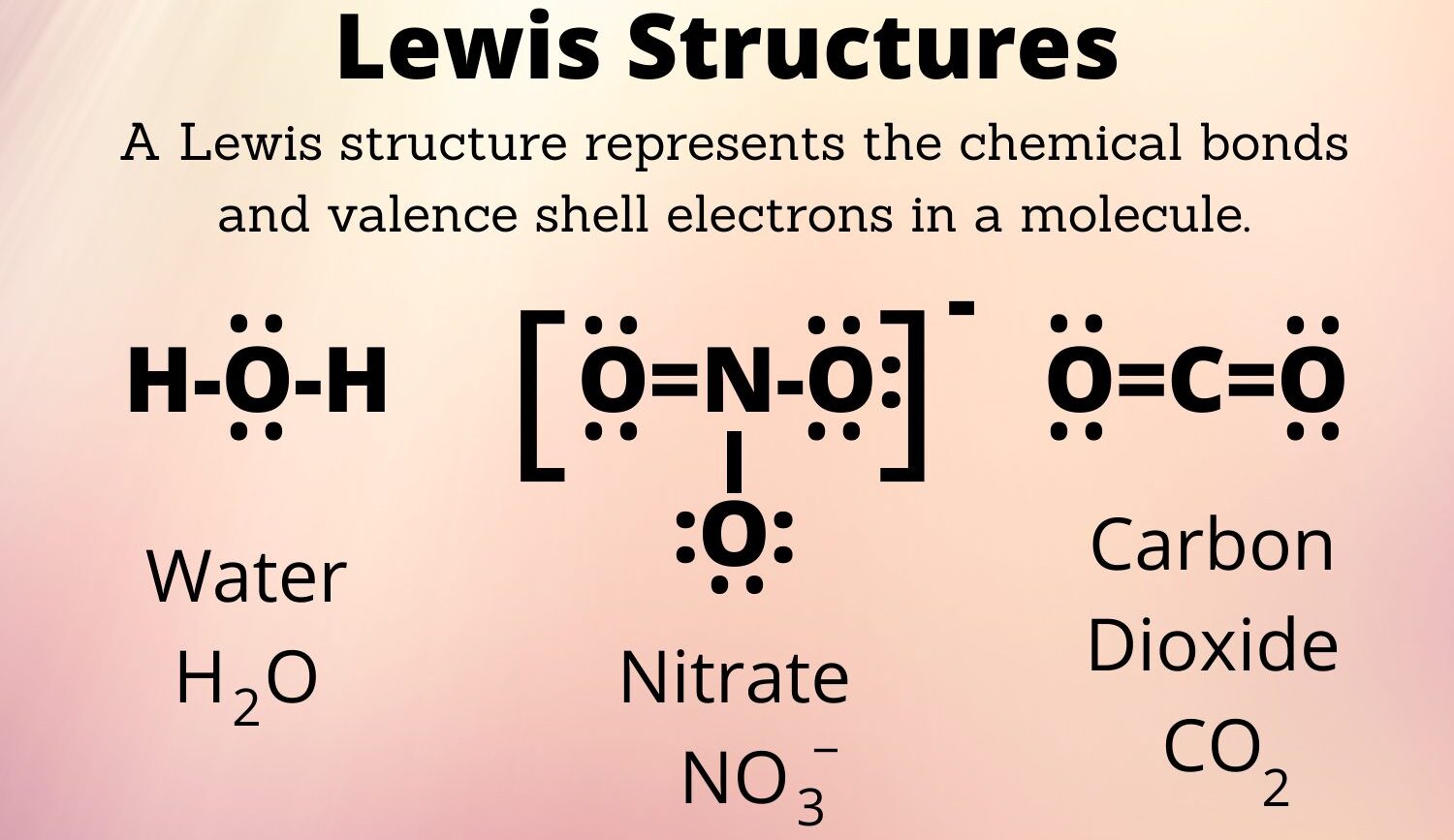
For many folks, chemistry can seem a bit, well, abstract, but Lewis structures, named after the brilliant chemist G.N. Lewis, help make these invisible atomic connections quite visible. They offer a simple, visual way to represent the valence electrons within a molecule or ion, showing us how atoms share or transfer electrons to achieve stability, so it's a very practical tool.
This guide will walk you through everything you need to know about the lewis structure of cn-, from the basic principles to the detailed steps for drawing it correctly. You'll get to see why the cyanide ion is structured the way it is, and we'll even touch on some interesting properties that come from this arrangement. It's a journey into the tiny world of electrons, really.
Table of Contents
- What Exactly is a Lewis Structure?
- The Basics of Valence Electrons
- Why is CN- So Interesting? (Cyanide Ion Lewis Structure)
- Step-by-Step Guide: Drawing the Lewis Structure for CN-
- Step 1: Count Total Valence Electrons
- Step 2: Choose the Central Atom
- Step 3: Connect Atoms with Single Bonds
- Step 4: Distribute Remaining Electrons
- Step 5: Form Multiple Bonds (if needed)
- Step 6: Check Formal Charges
- Understanding Formal Charges in CN-
- Calculating Formal Charge
- The Most Stable CN- Lewis Structure
- Common Questions About the Cyanide Ion
- What makes CN- a strong nucleophile?
- Is the CN- bond polar or nonpolar?
- How does the negative charge affect CN- reactivity?
What Exactly is a Lewis Structure?
A Lewis structure is, in essence, a shorthand drawing that shows how atoms bond in a molecule or ion. It helps us picture the valence electrons, which are the outermost electrons involved in chemical bonding, and how they are arranged around each atom. This visual representation, developed by G.N. Lewis, helps us understand molecular geometry and reactivity, you know, which is pretty neat.
These structures are built on the idea that atoms want to achieve a stable electron configuration, typically by having eight valence electrons, often called an "octet." Hydrogen, though, is a bit different; it only aims for two electrons. Atoms reach this stable state by sharing electrons in covalent bonds or, in some cases, by transferring them to form ionic bonds. So, it's all about reaching that happy, stable electron count, apparently.
The Basics of Valence Electrons
Valence electrons are the true stars of any Lewis structure. They're the electrons in the outermost shell of an atom, and they're the ones that get involved in forming chemical bonds. Knowing how many valence electrons each atom brings to the table is the very first thing you need to figure out when drawing a Lewis structure, that's really important.
- Jonathan Bailey Andrew Scott
- Does Simone Biles Mom Have Diabetes
- Exactly E Sextape
- Lucy Liu Height
- Tana Mongeau Leak
You can usually find this number by looking at the atom's group number on the periodic table. For example, carbon is in Group 14, so it has four valence electrons. Nitrogen, in Group 15, has five. These numbers are, like, your starting point for everything else, and it's a fairly simple way to begin.
Why is CN- So Interesting? (Cyanide Ion Lewis Structure)
The cyanide ion, CN-, is a fascinating little chemical species, and understanding its Lewis structure is quite revealing. It's made up of a carbon atom and a nitrogen atom, carrying an overall negative charge. This negative charge means there's an extra electron floating around that needs to be accounted for in the structure, which is a bit of a twist.
Cyanide is known for being toxic, but it's also a very useful building block in organic chemistry, so it's got a dual nature. Its unique reactivity comes directly from how its electrons are arranged, particularly the presence of that negative charge and the way the atoms bond. So, getting this structure right helps explain a lot about its behavior, really.
Step-by-Step Guide: Drawing the Lewis Structure for CN-
Drawing the lewis structure of cn- is a systematic process. If you follow these steps, you'll get it right every time, you know, which is helpful. It's a bit like following a recipe, where each step builds on the last, and it's not too difficult once you get the hang of it.
Step 1: Count Total Valence Electrons
This is where it all begins. First, you need to count the valence electrons for each atom in the ion. Carbon (C) is in Group 14, so it has 4 valence electrons. Nitrogen (N) is in Group 15, meaning it has 5 valence electrons. Now, don't forget that negative charge on the ion, because that means we add one extra electron to our count. So, in total, we have 4 (from C) + 5 (from N) + 1 (for the negative charge) = 10 valence electrons. That's the sum we need to work with, basically.
Step 2: Choose the Central Atom
In molecules with more than two atoms, you'd pick the least electronegative atom as the central one. However, for a diatomic ion like CN-, there isn't really a "central" atom in the usual sense. Both carbon and nitrogen are directly bonded to each other. So, this step is a bit simpler here; you just know they'll be connected, you know.
Step 3: Connect Atoms with Single Bonds
Now, you draw a single bond between the carbon and nitrogen atoms. A single bond represents two shared electrons. So, if we started with 10 total valence electrons and used 2 for this bond, we now have 10 - 2 = 8 electrons left to distribute. That's what we've got remaining, you see.
Step 4: Distribute Remaining Electrons
Next, you place the remaining electrons as lone pairs on the outer atoms first, making sure each atom achieves an octet if possible. Since there are only two atoms here, we just distribute them between carbon and nitrogen. Let's start by giving nitrogen, which is more electronegative, as many as it can take to get to eight. If nitrogen has one bond (2 electrons), it needs 6 more electrons to complete its octet. So, we place 3 lone pairs on nitrogen. We had 8 electrons left, and we used 6, leaving us with 2 electrons. These last 2 electrons must go on the carbon atom as a lone pair. So, carbon now has 2 electrons from the bond and 2 from its lone pair, making 4 electrons around it, while nitrogen has 2 from the bond and 6 from its lone pairs, making 8 electrons. Carbon is not happy yet, though, is it?
Step 5: Form Multiple Bonds (if needed)
At this point, carbon only has four electrons around it (two from the bond, two from its lone pair), which is not an octet. Nitrogen, however, has an octet. To give carbon its octet, we need to move some lone pair electrons from nitrogen to form more bonds between carbon and nitrogen. If we move one lone pair from nitrogen to form a double bond, carbon will have 6 electrons (4 from the double bond, 2 from its lone pair), and nitrogen will still have 6 (4 from the double bond, 2 from its remaining lone pair). Still not quite there, are we?
So, let's try moving *another* lone pair from nitrogen. If we move two lone pairs from nitrogen, we form a triple bond between carbon and nitrogen. Now, carbon has 6 electrons from the triple bond and 2 from its lone pair, totaling 8 electrons! Nitrogen has 6 electrons from the triple bond and 2 from its *one* remaining lone pair, also totaling 8 electrons! Both atoms now have an octet, which is really good. So, the most likely structure has a triple bond between carbon and nitrogen, with one lone pair on carbon and one lone pair on nitrogen, actually.
Step 6: Check Formal Charges
This final step is super important to confirm you have the most plausible Lewis structure. Formal charges help us decide which arrangement of electrons is best when multiple structures are possible, you know. We'll calculate the formal charge for each atom. The goal is to have formal charges as close to zero as possible, and for any negative charge on an ion to reside on the more electronegative atom, which is typically what you want.
Understanding Formal Charges in CN-
Formal charges are a bit like accounting for electrons. They help us determine the most stable Lewis structure for a molecule or ion. It's a way to see if electrons are distributed fairly, or if one atom is carrying too much of a positive or negative burden, so it's a very useful concept.
Calculating Formal Charge
The formula for formal charge is: Formal Charge = (Valence Electrons) - (Non-bonding Electrons) - (1/2 * Bonding Electrons)
Let's apply this to our triple-bonded CN- structure:
- For Carbon (C):
- Valence electrons: 4
- Non-bonding electrons (lone pair): 2
- Bonding electrons (triple bond): 6
- Formal Charge (C) = 4 - 2 - (1/2 * 6) = 4 - 2 - 3 = -1
- For Nitrogen (N):
- Valence electrons: 5
- Non-bonding electrons (lone pair): 2
- Bonding electrons (triple bond): 6
- Formal Charge (N) = 5 - 2 - (1/2 * 6) = 5 - 2 - 3 = 0
So, the carbon atom has a formal charge of -1, and the nitrogen atom has a formal charge of 0. The sum of the formal charges (-1 + 0) equals the overall charge of the ion, which is -1. This is a good sign that our structure is correct, you know, and it's what we expect.
The Most Stable CN- Lewis Structure
Our calculation shows that the carbon atom carries the negative formal charge in the triple-bonded structure. You might think the negative charge should be on nitrogen, since nitrogen is more electronegative than carbon. However, a structure where nitrogen has the negative charge (e.g., C-N with a single bond and more lone pairs on N) would violate the octet rule for carbon, or result in larger formal charges, which is not ideal. So, the triple-bonded structure with the negative charge on carbon is actually the most stable and correct representation, even if it seems a little counterintuitive at first glance. It's a subtle point, but an important one, really.
Common Questions About the Cyanide Ion
People often have questions about the cyanide ion, CN-, beyond just drawing its Lewis structure. Its properties are quite interesting, and they often relate back to how its electrons are arranged, so it's worth exploring these things.
What makes CN- a strong nucleophile?
The cyanide ion is, in fact, a very strong nucleophile. This means it's really good at donating its electron pair to form new bonds with electron-deficient atoms, which are called electrophiles. The negative charge on the carbon atom, as we saw in the Lewis structure, makes that carbon particularly electron-rich and ready to share. This high electron density makes it very attractive to positive centers, so it's quite reactive in that way.
Is the CN- bond polar or nonpolar?
The bond between carbon and nitrogen in CN- is definitely polar. Nitrogen is more electronegative than carbon, meaning it pulls the shared electrons in the triple bond a bit closer to itself. Even though the overall ion has a negative charge, there's an uneven distribution of electron density within the bond itself, creating a partial negative charge near the nitrogen and a partial positive charge near the carbon. So, yes, it's a polar covalent bond, you know.
How does the negative charge affect CN- reactivity?
The overall negative charge on the cyanide ion significantly impacts its reactivity. This extra electron makes the ion much more willing to participate in reactions where it can donate an electron pair, as we discussed with its nucleophilic character. It also means it's more attracted to positively charged species or regions of low electron density, which drives many of its chemical interactions. So, that single extra electron really changes things, doesn't it?
To learn more about basic chemical bonding on our site, and to explore other related concepts, you can also check out this page understanding molecular geometry.
For more detailed information on Lewis structures and their applications, you might find it helpful to visit a general chemistry resource like LibreTexts Chemistry, which offers a broad range of topics. They have a lot of good stuff, actually.
- Chris Christie Speaking Fee
- симонетта стефанелли
- Brigitte Macron Wedding Dress
- Gabriel Fernandez In Hospital Bed
- Bridget Wilson


Detail Author:
- Name : Joseph Mayert III
- Username : eemard
- Email : april.wilkinson@mann.org
- Birthdate : 1993-10-26
- Address : 40156 Kub Tunnel Suite 977 Dellville, TX 96684
- Phone : 1-463-817-0708
- Company : Pagac Group
- Job : Welding Machine Setter
- Bio : Fuga sapiente maiores et sit. Ducimus placeat repellat eos beatae aut ut. Non sed iusto maxime autem in magnam voluptas.
Socials
facebook:
- url : https://facebook.com/rex_xx
- username : rex_xx
- bio : Dignissimos id nam laudantium est ab reprehenderit.
- followers : 5554
- following : 956
linkedin:
- url : https://linkedin.com/in/rex3173
- username : rex3173
- bio : Veniam vel quod nulla qui.
- followers : 5749
- following : 1640
instagram:
- url : https://instagram.com/rexwalter
- username : rexwalter
- bio : Quo omnis quisquam aut ea laborum nemo est. Reiciendis debitis sed sunt itaque consequatur.
- followers : 1422
- following : 2479