When we hear the name "Lewis," a lot of different pictures might pop into our heads, you know? Perhaps you think of a race car driver making strategic choices about downforce for better tire wear, or maybe a lively radio host chatting with listeners daily on SiriusXM's Radio Andy, talking about a new house or having a kiki. Some might even picture a character from a video game, or ponder deep philosophical arguments about divinity. But today, we're talking about a very specific kind of Lewis – one who helped us see the unseen world of atoms and how they bond together.
We're going to explore the Lewis structure of the cyanide ion, CN-, which is a pretty fascinating little particle with a big story to tell about how atoms connect. This isn't about someone running a higher downforce rearwing, or whether a Lewis gun is superior to an M60, or even the regrets of attending a particular university. No, this is about Gilbert N. Lewis, a chemist whose ideas, arguably, made it much easier for us to draw out how atoms share their electrons.
Understanding these structures, you see, is a fundamental step in chemistry. It helps us guess how a molecule might behave, how it might react, and what its overall shape could be. So, let's peel back the layers and really get to grips with what makes the CN- ion tick, from its individual atoms to its final, stable arrangement, actually.
- Lisa Left Eye Net Worth
- Brittany Nicole Holbrook
- Sexy Videocom
- Britney Theriot Children
- Best Remote Iot Vpc Network Raspberry Pi
Table of Contents
- What Even Is a Lewis Structure, Anyway?
- Gathering Our Atoms: Carbon, Nitrogen, and That Extra Electron
- Putting Them Together: The Core of CN-
- Making Sure Everyone is Happy: The Octet Rule
- Formal Charges: Checking the Electron "Fairness"
- Is There Another Way? Exploring Resonance for CN-
- What CN- Does in the Real World: Beyond the Diagram
- Common Questions About the Cyanide Ion's Structure
- Bringing It All Together: Why This Matters
What Even Is a Lewis Structure, Anyway?
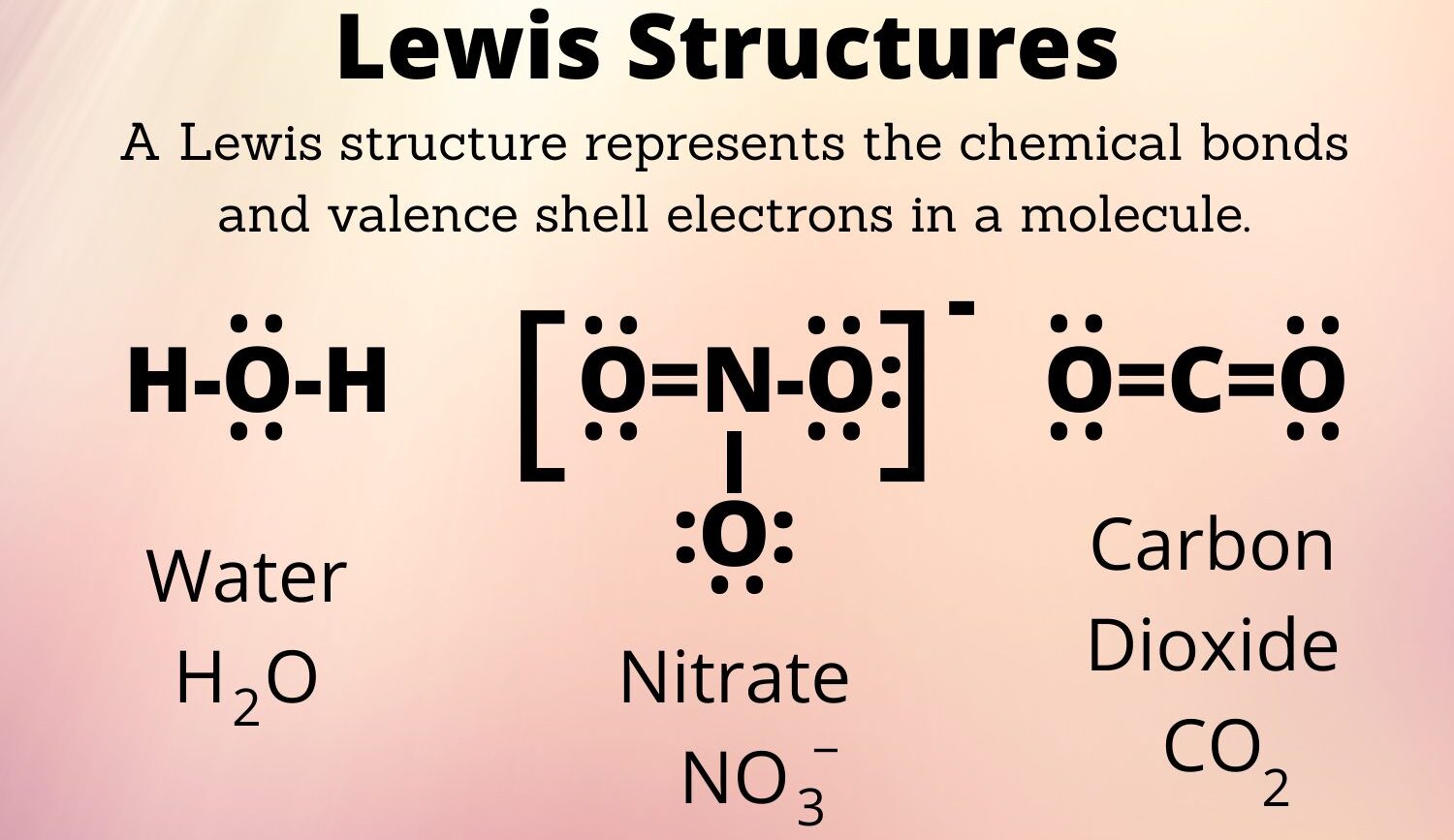
A Lewis structure, sometimes called a Lewis dot diagram, is basically a way to draw how atoms in a molecule or ion share their outermost electrons, you know, the ones involved in making connections. It's a visual shorthand that helps us understand the arrangement of these electrons, which are often called valence electrons, in some respects.
The main point of these drawings is to show every atom and all its valence electrons, whether they are shared in a bond or sitting alone as what we call "lone pairs." This helps us predict the stability of a molecule and how it might act, which is pretty useful, actually.
Gathering Our Atoms: Carbon, Nitrogen, and That Extra Electron
To start drawing the Lewis structure for CN-, we first need to get to know our players: carbon and nitrogen. Each atom brings a certain number of valence electrons to the table, and we also have to remember that little minus sign, which means an extra electron is joining the party, so to speak.
- Kat Marie Alex Adam
- Bridgette Wilson Sampras
- фелисити хаффман побрилась налысо
- Richard Boone Net Worth
- Yesjulz Leaks
Carbon, for instance, is a really versatile atom, known for forming four bonds. It sits in Group 14 of the periodic table, which means it has four valence electrons. Nitrogen, on the other hand, is in Group 15, so it comes with five valence electrons, giving it a bit of a different personality, you know.
The negative charge on CN- means that this ion has one more electron than the total sum of valence electrons from a neutral carbon and a neutral nitrogen atom. This extra electron is super important for our counting, as a matter of fact.
Counting Up the Valance Electrons for CN-
Let's do the math to figure out the total number of valence electrons we need to place in our Lewis structure. This step is pretty crucial, because if we get this wrong, everything else will be off, too.
Carbon contributes 4 valence electrons. Nitrogen brings 5 valence electrons. Then, that negative charge adds 1 more electron to our count. So, when you add it all up, we have a grand total of 4 + 5 + 1 = 10 valence electrons to work with for the CN- ion, okay.
Putting Them Together: The Core of CN-
Now that we know how many electrons we have, we can start arranging the atoms. Since there are only two atoms, carbon and nitrogen, the central atom choice is pretty straightforward – they're just connected to each other, you know. There's no "central" atom in the usual sense with just two.
We always start by placing a single bond between the atoms. This single bond uses up two of our total valence electrons. So, we draw a line connecting C and N, and we now have 10 - 2 = 8 electrons left to distribute, you see.
Distributing the Remaining Electrons
With our 8 remaining electrons, we need to make sure each atom gets as close to a full outer shell as possible. For most atoms, this means having eight electrons around them, which is often called the octet rule. We usually start by giving lone pairs to the more electronegative atom first, which is nitrogen in this case, actually.
Nitrogen already has two electrons from the single bond. To get to eight, it needs six more. So, we place three lone pairs (six electrons) on the nitrogen atom. This leaves us with 8 - 6 = 2 electrons still to place, as a matter of fact.
These last two electrons must go on the carbon atom, since nitrogen now has its octet. So, carbon gets one lone pair. At this point, carbon has two electrons from the single bond plus its one lone pair, making a total of four electrons around it, which is not an octet yet, you know.
Making Sure Everyone is Happy: The Octet Rule
Right now, nitrogen looks happy with its eight electrons (two from the bond, six from lone pairs). Carbon, however, only has four electrons around it (two from the bond, two from its lone pair). This is where we need to get creative to make everyone satisfied, basically.
When an atom doesn't have a full octet, it can often share more electrons with its bonding partner to form multiple bonds. In this situation, carbon needs four more electrons to reach its octet. Nitrogen has three lone pairs it could share, so it's a good candidate to help out, you see.
If nitrogen shares one of its lone pairs with carbon, they form a double bond. Carbon would then have six electrons (four from the double bond, two from its lone pair). Still not an octet for carbon. If nitrogen shares another lone pair, they form a triple bond. Now, carbon would have eight electrons (six from the triple bond, two from its lone pair), and nitrogen would still have eight (six from the triple bond, two from its remaining lone pair). This looks pretty good, actually.
The Triple Bond: A Strong Connection
So, the most stable Lewis structure for CN- features a triple bond between the carbon and nitrogen atoms. This triple bond means they are sharing six electrons in total, which is a pretty strong connection, you know. On the carbon atom, there will be one lone pair of electrons, and on the nitrogen atom, there will also be one lone pair of electrons.
To visualize this, you'd draw a 'C' with two dots next to it (representing the lone pair), then three lines connecting to an 'N' with two dots next to it (representing its lone pair). Then, you'd put square brackets around the whole thing with a negative sign outside to show it's an ion, which is pretty standard, you know.
Formal Charges: Checking the Electron "Fairness"
Formal charges help us figure out if the electrons are distributed in the "best" possible way within a Lewis structure. It's like a bookkeeping tool to see how many electrons an atom "owns" compared to how many it starts with as a neutral atom. The goal is often to have formal charges as close to zero as possible, and for any negative charge to be on the more electronegative atom, generally speaking.
The formula for formal charge is: (Valence electrons of the neutral atom) - (Non-bonding electrons) - (1/2 * Bonding electrons). Let's apply this to our CN- structure. Remember, we have a C-N triple bond, with one lone pair on C and one lone pair on N, you see.
For Carbon: It starts with 4 valence electrons. In our structure, it has 2 non-bonding electrons (from its lone pair) and 6 bonding electrons (from the triple bond). So, its formal charge is 4 - 2 - (1/2 * 6) = 4 - 2 - 3 = -1. That's a negative one charge on the carbon, as a matter of fact.
For Nitrogen: It starts with 5 valence electrons. In our structure, it has 2 non-bonding electrons (from its lone pair) and 6 bonding electrons (from the triple bond). So, its formal charge is 5 - 2 - (1/2 * 6) = 5 - 2 - 3 = 0. Nitrogen has a formal charge of zero, which is pretty good, you know.
The sum of the formal charges (-1 + 0 = -1) matches the overall charge of the cyanide ion, which is also -1. This tells us our electron counting and placement are correct for this particular structure, so that's a good sign, really.
Why Formal Charges Help Us Understand
Formal charges are super helpful because they guide us toward the most plausible Lewis structure when multiple arrangements might seem possible. A structure with formal charges closer to zero on most atoms is generally more stable. Also, if there is a negative charge, it's usually better for it to be on the atom that likes electrons more (the more electronegative atom), as a matter of fact.
In the case of CN-, placing the negative formal charge on carbon might seem a bit odd at first, since nitrogen is more electronegative. However, this particular arrangement with the triple bond and the -1 on carbon is actually the most stable and widely accepted Lewis structure for the cyanide ion, basically. This is because it allows both atoms to achieve an octet, which is a very strong driving force for stability, you know.
Is There Another Way? Exploring Resonance for CN-
Sometimes, a single Lewis structure just isn't enough to fully describe a molecule or ion. When electrons can be arranged in more than one valid way, we talk about resonance structures. These are different drawings that show the same connectivity but with different placements of multiple bonds and lone pairs, you see. The actual molecule is then thought of as a blend, or a hybrid, of all these possible structures, more or less.
For the cyanide ion, CN-, the structure we just drew with the triple bond between carbon and nitrogen, one lone pair on carbon, and one lone pair on nitrogen, is the primary and most stable one. Could there be other ways to draw it? Well, yes, but they are typically much less favorable, you know.
For example, you could imagine a structure with a double bond between C and N. If you did that, carbon would need more lone pairs, and nitrogen would have more lone pairs. Let's say C=N with two lone pairs on C and two lone pairs on N. Let's check the formal charges there. Carbon: 4 - 4 - (1/2 * 4) = 4 - 4 - 2 = -2. Nitrogen: 5 - 4 - (1/2 * 4) = 5 - 4 - 2 = -1. The total charge is -3, which doesn't match the ion's -1 charge. So, this one is clearly not right, actually.
What about a single bond? C-N. Carbon would have three lone pairs (6 electrons), Nitrogen would have three lone pairs (6 electrons). Formal charges: Carbon: 4 - 6 - (1/2 * 2) = 4 - 6 - 1 = -3. Nitrogen: 5 - 6 - (1/2 * 2) = 5 - 6 - 1 = -2. Total charge -5. Again, not matching. This shows us that the triple bond structure is, in fact, the most fitting one for CN-, which is pretty neat.
The key takeaway here is that while resonance is a powerful idea, not every molecule will have multiple significant resonance forms. For CN-, the triple-bonded structure with the negative charge on carbon is, arguably, the best representation because it satisfies the octet rule for both atoms and keeps the overall charge correct, you know.
What CN- Does in the Real World: Beyond the Diagram
Understanding the Lewis structure of CN- isn't just an academic exercise; it helps us grasp why this ion behaves the way it does in actual chemical situations. The presence of that lone pair on carbon and the triple bond makes it a very interesting and reactive species, as a matter of fact.
Because carbon has a lone pair and a negative formal charge, the cyanide ion is what chemists call a strong nucleophile. This means it really likes to "attack" parts of other molecules that are lacking electrons, making it a key player in many organic reactions. It's also a powerful ligand, which means it can form strong connections with metal ions, which is quite important in coordination chemistry, you know.
Its linear shape, dictated by the two atoms and the triple bond, also influences how it interacts with other substances. So, the simple Lewis diagram, in a way, gives us a window into its much larger chemical story, which is pretty cool, actually.
Common Questions About the Cyanide Ion's Structure
People often have questions when they first learn about the Lewis structure of CN-. Here are a few common ones, and some straightforward answers, you know.
Is CN- linear or bent?
The cyanide ion, CN-, is linear. With only two atoms, and a triple bond connecting them, there's no other way for them to arrange themselves. It's a straight line, basically.
Why does carbon have the negative formal charge in CN-?
Even though nitrogen is more electronegative, the most stable Lewis structure for CN- ends up with a negative formal charge on carbon. This happens because it allows both carbon and nitrogen to achieve a full octet of electrons, which is a very strong factor in determining stability for these atoms, as a matter of fact. It's a bit of a trade-off that works out best for the whole ion, you know.
How many lone pairs are on the carbon atom in CN-?
In the most accepted Lewis structure for CN-, there is one lone pair of electrons on the carbon atom. This lone pair, along with the triple bond, gives carbon its full octet, you see.
Bringing It All Together: Why This Matters
So, we've walked through the steps for drawing the Lewis structure of the cyanide ion, CN-. We started by counting all the valence electrons, then connected the atoms with a single bond, and distributed the remaining electrons. We saw how forming a triple bond was necessary to make sure both carbon and nitrogen had their full octets, and then we checked our work using formal charges, which is pretty important, you know.
Understanding these structures, like the one for CN-, is a cornerstone of chemistry. It helps us predict how molecules will look and how they might react, which is super useful for anyone studying or working with chemicals. It’s like learning the alphabet before you can read a book, in a way. If you're keen to keep exploring how atoms connect, you can learn more about valence electrons on our site, and perhaps link to this page understanding chemical bonds for more ideas, you know.
Keep practicing these kinds of drawings, because the more you do, the clearer the patterns become. It really helps to build a strong foundation in chemistry, as a matter of fact. For further reading on fundamental chemistry principles, you might find resources from the International Union of Pure and Applied Chemistry (I
- Gloria Loring
- Aroomikim Onlyfans Leaks
- Remoteiot Web Ssh Example Android
- Boiiy4u
- Dick Van Dyke Worth


Detail Author:
- Name : Prof. Caitlyn Lindgren IV
- Username : dietrich.brown
- Email : rylan.runte@yahoo.com
- Birthdate : 1998-05-07
- Address : 8574 Ruthie Islands Noemyburgh, GA 31502
- Phone : 743-286-9233
- Company : Lehner, Little and Skiles
- Job : Tree Trimmer
- Bio : Qui aut blanditiis a qui unde consectetur excepturi. A tempora delectus eum qui. Cumque vitae in illum ex quisquam adipisci doloremque.
Socials
tiktok:
- url : https://tiktok.com/@mariane_hudson
- username : mariane_hudson
- bio : Dolor ut commodi minima. Aspernatur et vel laborum libero fugit.
- followers : 2455
- following : 87
facebook:
- url : https://facebook.com/hudsonm
- username : hudsonm
- bio : Inventore assumenda perferendis ab sit non est in.
- followers : 2120
- following : 2075
linkedin:
- url : https://linkedin.com/in/mhudson
- username : mhudson
- bio : Porro perferendis quis dicta minima et atque et.
- followers : 5639
- following : 2556
twitter:
- url : https://twitter.com/mariane_official
- username : mariane_official
- bio : Deserunt omnis consectetur veniam ab quos sint. Debitis repellat molestiae qui delectus qui temporibus totam. Et nulla nostrum quae recusandae assumenda qui.
- followers : 1347
- following : 829
instagram:
- url : https://instagram.com/mhudson
- username : mhudson
- bio : Quae aut in et explicabo quis. Sit iusto id magnam optio sequi quis.
- followers : 3692
- following : 2457